MicrosensDx announces a portable coronavirus testing kit for research use that takes less than 25 minutes from taking a sample to test result
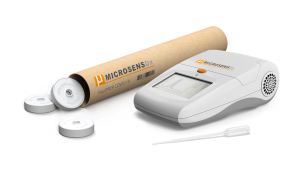 The MicrosensDx RapiPrep® system enables highly sensitive SARS-CoV-2 detection in less than 25 minutes from taking a sample to test result. The portable, desktop kit will be used initially in scientific laboratories involved in vaccine and therapeutic research and development.
The MicrosensDx RapiPrep® system enables highly sensitive SARS-CoV-2 detection in less than 25 minutes from taking a sample to test result. The portable, desktop kit will be used initially in scientific laboratories involved in vaccine and therapeutic research and development.MicrosensDx is now engaged in a programme to gain CE accreditation for the SARS-CoV-2 test which will permit its sale and use in the diagnosis of the COVID-19 disease. The CE-marked kits will provide rapid ‘on demand’ testing support alongside the large-scale hospital laboratory testing instruments and will also allow disease testing to take place in facilities such as clinics, doctor’s offices and in the workplace.
The key features of the RapiPrep® SARS-CoV-2 research use test are:
- Any sample type is suitable: Including virus-containing culture media and tissue, clinical samples such as throat, nasal, sputum, faeces.
- Rapid detection time: Less than 25 min to result.
- Detection method: Detection of the viral RNA genome based on the MicrosensDx modified LAMP nucleic acid amplification technology, which is already in routine use in the company’s tuberculosis and norovirus clinical diagnostics products.
- Accuracy: The RapiPrep® system incorporates the company’s proven magnetic bead-based virus extraction technology that gives enhanced sensitivity over other testing methods.
- Reliability: The modified LAMP method incorporates procedure controls to prevent false negatives, which will be of critical importance in the future CE-marked test.
- Safety: Immediate virus inactivation takes place inside the test cartridge.
- Easy to operate: Three simple steps, suitable for semi-skilled operators.
- Automated detection: The instrument has visual representation of test result and confirmation that the procedure control has run successfully.
The RapiPrep® system will be undergoing clinical assessment imminently within the UK to demonstrate its value as a rapid COVID-19 screening tool.
MicrosensDx CEO, Dr Christopher Stanley, commented: “Our RapiPrep® system will enable rapid, sensitive and specific detection of SARS-CoV-2 by the research community and when CE-marked will permit highly effective screening of patient samples in hospitals and in the community. This will allow more informed decision-making on disease containment and delay methods, whether that be treatment or self-isolation. We are confident that this technology when launched for clinical use can make a significant impact on flattening the outbreak curve and reducing the enormous burden on the healthcare system.
---------------------------------------------------------------------------------------------------------------------------
MicrosensDx
With more than eighteen years of experience in addressing unmet needs in diagnostic markets, MicrosensDx specialises in developing cost-effective, easy to use and easily automatable technologies for the extraction and detection of intact pathogens and pathogen nucleic acids from the widest range of samples. The company has been highly successful in developing a laboratory test for the ‘mad cow’ agent BSE and with its US partner now dominates the worldwide market for prion disease testing. In its tuberculosis (TB) detection programme MicrosensDx is using the company’s RapiPrep® technology in products intended for patient-side testing in local clinics in developing countries where rapid disease detection is needed. The company undertakes clinical trials in South Africa and also sells simplified TB testing products in several markets worldwide, including achieving major sales in Japan. In the field of RNA virus testing MicrosensDx has a long running programme to develop rapid tests in collaboration with a UK hospital and now has utilised its expertise to develop a sensitive detection product for SARS-CoV-2.
Contact: Claire Thompson on behalf of MicrosensDx
claire.thompson@agilityhealthtech.com / +44 (0)7885 591 187 / www.microsensdx.com





















