How Early Notified Body Opinion Strategy Prevents Delays in Combination Products
How Early Notified Body Opinion Strategy Prevents Article 117 Delays in Combination Products
Most delays are not caused by regulation – they are caused by strategy and timing.
Early Notified Body Opinion (NBOp) strategy is one of the most effective ways to prevent Article 117 delays and protect EU launch timelines for combination products. For drug-led combination products in the EU, the Notified Body Opinion is often treated as a late-stage regulatory requirement. In reality, it’s a critical development milestone that must be planned from the outset — not only to avoid delays, but to optimise the performance of the drug-device system for patients.

Figure 1. Notified Body Opinion Mindset
Why This Matters
The European Union remains one of the most important and complicated markets for combination products. Early alignment of drug and device development not only reduces delays but strengthens therapeutic performance and improves patient usability.
With the introduction of the Medical Device Regulation (EU MDR) and Article 117 for products where the medicinal product is the primary mode of action, companies must:
Obtain a Notified Body assessment of the device component, and
Include the Notified Body Opinion within the EMA submission
This creates a fundamentally sequential pathway:
Notified Body assessment (device compliance)
EMA review (including NB Opinion)
While the EU pathway is structured sequentially and the US pathway is more integrated, both ultimately assess the performance of the combined drug-device system. The difference between EU and US pathways is not just procedural — it fundamentally impacts development timelines and evidence generation. Figure 2 illustrates the difference between the EU sequential pathway and the US integrated review.
The key implication: Your device evidence – and the underlying device-drug system performance must be ready months before your EMA submission.
This is where many programmes encounter avoidable delays — and, more importantly, risk suboptimal system performance if not addressed early.
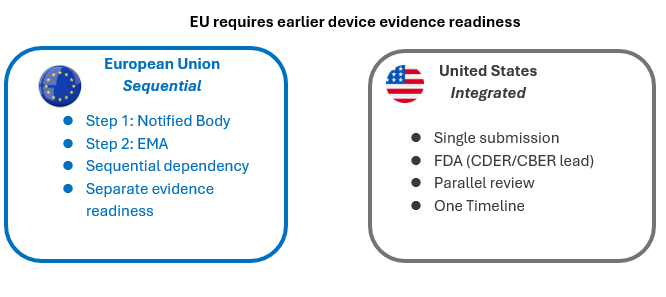
Figure 2. Comparison of Regulatory Pathways for Drug-Led Combination Products in the EU and the US
The Real Challenge
Most companies still develop combination products primarily with the US in mind — although this is increasingly shifting as EU pathways evolve.
In practice, this creates a recurring pattern across programmes:
GSPR (General Safety and Performance Requirements) mapping starts too late
Device development and supporting evidence are incomplete or misaligned
Critical gaps are identified just before submission
The result is not just regulatory friction — it reflects underlying gaps in system-level development and can lead to programme disruption.
Where Things Go Wrong
Most Notified Body challenges are not caused by regulatory complexity, but by gaps in development strategy and timing. At the core of the Notified Body Opinion is compliance with Annex I of EU MDR—the General Safety and Performance Requirements (GSPRs), which define the expectations for safety, design, manufacturing, and information provided with the device. Figure 2 provides a high-level overview of the GSPR framework.
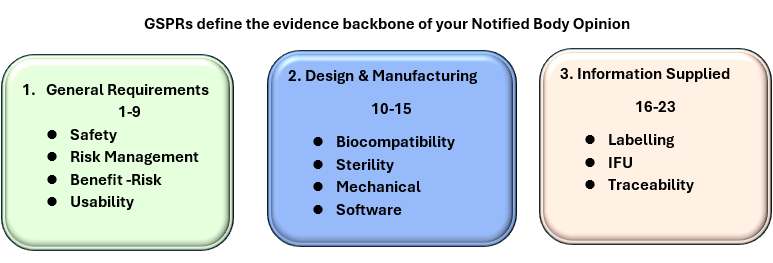
Figure 3. Overview of General Safety and Performance Requirements ( Annex I, EU MDR)
These are not new concepts. However, under MDR, expectations are more explicit, structured, and scrutinised.
A robust NBOp requires:
Clear mapping of all applicable GSPRs
Justification for applicability (or non-applicability)
Supporting evidence generated at the right time
Commercial strategy in alignment with the three-pillar operating model and associated reimbursement – being allowed to place on the market is not the same as selling it
The science is the same for FDA and EU; the difference is structure and timing, and strong programmes leverage both to build better products
Common pitfalls we see in practice:
Assuming FDA evidence is sufficient; it often is not aligned to GSPR expectations.
The science for FDA and EU is the same; the structure and timing are not — and that’s where programmes run into trouble.
Treating NBOp as a checklist exercise instead of integrating it into development.
Starting too late after design decisions are already locked.
Underestimating evidence requirements. Particularly for areas like usability, biocompatibility, material requirements, risk management, process validation, stability, clinical evaluation, etc.
Evidence expectation from Notified Bodies can also be less predictable than many teams anticipate.
This could be particularly true for areas such as:
Process validation
Stability (including device–drug interaction considerations)
Sterilisation
Clinical evaluation of the device component
Without early clarity, teams often fail to perform the necessary development work or generate evidence that is insufficient, misaligned, or requires rework. This leads to avoidable delays and, more critically, poor optimised product performance.
The Impact
When NBOp planning isn’t integrated into the therapeutic-system design, the consequences are significant:
Delays of months — or even years
Costly redesign or additional testing
Increased regulatory questions
Strain on cross-functional teams
Missed market opportunities
For pharmaceutical products, timing is directly linked to asset value and market opportunity – but only when supported by robust science and well-designed product systems. Delays in EU access can have substantial commercial implications.
Most NBOp challenges are not regulatory – they are developmental and organisational
What Experienced Teams Do Differently
Teams that navigate NBOp successfully take a fundamentally different approach:
1. They treat NBOp as part of development — not a submission activity
GSPRs are considered from the concept stage, not retrofitted later — a shift that also helps bridge the common cultural gap between drug and device teams. Rather than following a linear, late-stage compliance approach, experienced teams integrate GSPRs throughout development. Figure 4 illustrates the shift from a traditional “waterfall” approach to a GSPR-integrated development mindset.
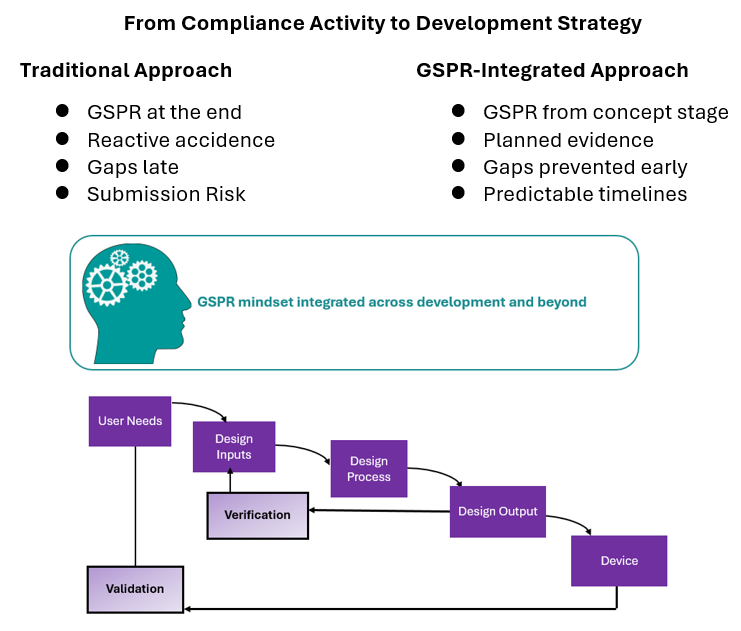
Figure 4. Integrating GSPRs into Product Development vs Traditional Waterfall Approach
2. They define evidence early
They understand:
What evidence is required
Why it is required
When it needs to be available
3. They align functions from day one
Regulatory, technical, and commercial teams work to a shared timeline and market strategy. See the Three Pillar Operating model for details.
4. They stay ahead of regulatory change
Active regulatory surveillance ensures strategy can adapt as expectations evolve.
5. They understand the purpose behind the requirements
GSPRs are not administrative — they are designed to ensure patient safety and product performance.
In practice, achieving this level of alignment and foresight often requires specialist expertise in both device and pharmaceutical regulatory expectations, particularly for complicated combination products.
Common Misconceptions
“These requirements are new.”
No, they aren’t. Many existed under the Essential Requirements framework — but MDR has increased the level of rigour and transparency.“It’s too complex to get a Notified Body Opinion.”
It is complex — but manageable with the right planning.“GSPRs are overly burdensome.”
Each requirement serves a purpose. The challenge is not the requirements themselves — it is how early they are considered.
A Shift in Mindset
For leaders and development teams, this is ultimately about mindset. Successful programmes are not built on reactive compliance. They are built on intentional, global planning.
This means:
Planning target markets early including EU NBOp requirements from the outset.
Embedding GSPRs into the Quality Management System.
Driving cross-functional alignment — Regulatory, technical, and commercial working together from day one — see Three pillar model for details.
Thinking globally — not sequentially or reactively. EU, Japan, South Korea and other markets should not be considered as “add-ons”.
Importantly, leaders should adopt a learning mindset across regions. Different health authorities often emphasise different aspects, although the underlying scientific expectations for safety and performance are largely consistent across regions. For example,
The EU may focus more explicitly on structured GSPR compliance
Other markets such as Japan, South Korea, China, etc. may bring additional perspectives on usability, quality systems, or device testing expectations
These are not just regulatory hurdles — they are opportunities to strengthen your product. Organisations that step back and ask, “What can we learn from each market?” are better positioned to:
Anticipate requirements earlier
Reduce rework
Ultimately design more robust, patient-centric products
How CPMD Consulting Ltd. Supports Successful NBOp Strategies
CPMD Consulting Ltd. is a specialist advisory firm focused on combination products and medical devices, supporting leadership teams in navigating complex global regulatory and development pathways.
Many organisations struggle with delays, evidence gaps, and misaligned timelines because Notified Body Opinion (NBOp) is often treated as a late-stage regulatory hurdle rather than a strategic milestone. CPMD Consulting Ltd. helps teams turn NBOp into an opportunity to strengthen products and streamline approvals.
We work with organisations to:
Identify evidence gaps early and align device and drug timelines
Interpret and apply GSPRs effectively to avoid last-minute surprises
Strengthen technical documentation and risk management
Improve cross-functional collaboration across regulatory, quality, technical, and commercial teams. See link to the Three Pillar Operating model for details.
Prepare confidently for Notified Body and EMA interactions
By integrating NBOp requirements early into strategy and execution, CPMD Consulting Ltd. helps organisations de-risk development, de-risk project launch timelines, and build more robust, patient-centric, globally aligned products — keeping the global standard of care in mind.
Contact and Resources
Website: www.cpmd.co.uk- expert guidance on strategic support for combination products.
Email: contact@cpmd.co.uk - reach out to discuss how CPMD Consulting Ltd. can support your NBOp strategy.
LinkedIn: Combination Product and Medical Device Consulting Ltd.- connect for updates and insights.
NBOp Preparation Checklist for Leaders
Download the comprehensive NBOp readiness checklist here:
Closing Perspective
Health authorities — whether in the EU, US, Japan, South Korea, Malaysia or elsewhere — are aligned on one fundamental goal which is to ensure safe, effective products for patients.
The frameworks may differ and evidence expectations may vary, but these differences are often complementary rather than conflicting.
Leaders who recognise this can shift — from managing regional differences to leveraging global consistency and moving from a compliance-driven approach to a product excellence mindset:
Leveraging insights across regions
Integrating the most stringent and relevant requirements early
Using global expectations to raise the quality of their product — not just meet minimum thresholds
For drug-led combination products in the EU, success still depends on planning early enough to ensure the right evidence and underlying scientific justification is available at the right time.
But the most effective organisations go one step further. They don’t just plan for compliance — they design with a global standard of care in mind.
Notified Body Opinion is not a checkbox. It is a development strategy.
Author: Combination Products and Medical Devices Consulting Ltd.
Editorial support: The blog was prepared with the assistance of FSI.
© 2026 CPMD Consulting | Combination Product and Medical Device Consulting Ltd. All rights reserved.
The views, frameworks, and strategic perspectives presented in this article are proprietary to CPMD Consulting Ltd. and may not be reproduced or used without written permission.

